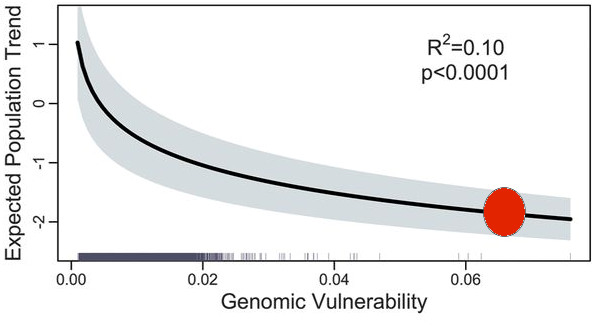
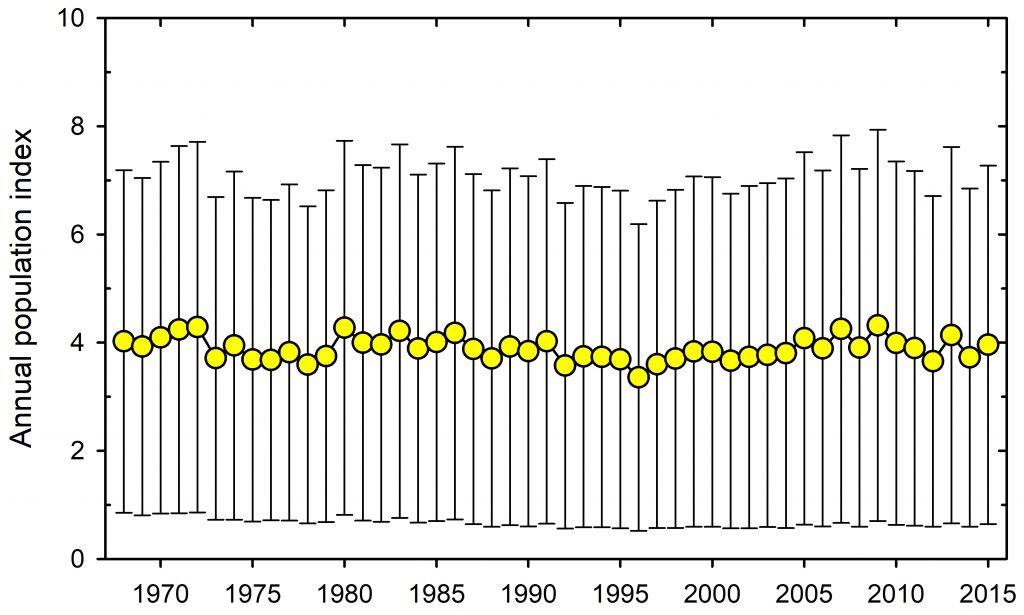
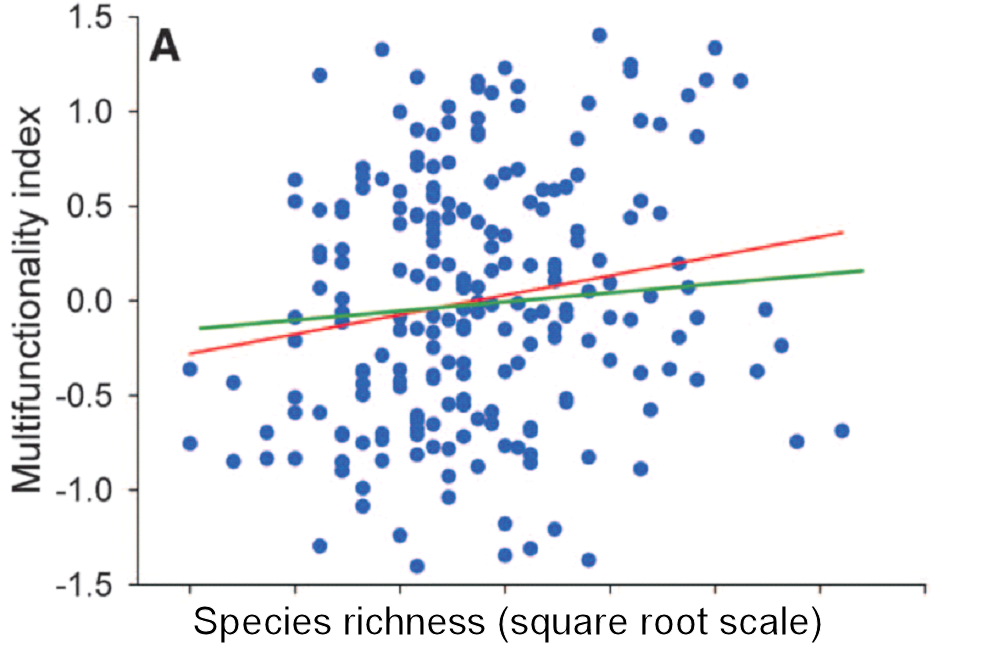
The loss of large mammals and birds in the Pleistocene was highlighted many years ago (Martin and Wright 1967, Grayson 1977, Guthrie 1984 and many other papers). Hypotheses about why these extinctions occurred were flying left and right for many years with no clear consensus (e.g. Choquenot and Bowman 1998). The museums of the world are filled with mastodons, moas, sabre-tooth tigers and many other skeletons of large mammals and birds long extinct. The topic has come up again in a discussion of these extinctions and a prognosis of future losses (Smith et al. 2018). I do not want to question the analysis in Smith et al. (2018) but I want to concentrate on this one quotation that has captured the essence of this paper in the media:
“Because megafauna have a disproportionate influence on ecosystem structure and function, past and present body size downgrading is reshaping Earth’s biosphere.”
(pg. 310).
What is the evidence for this very strong statement? The first thought that comes to mind is from my botanical colleagues who keep reminding me that plants make of 99% of the biomass of the Earth’s ecosystems. So, if this statement is correct, it must mean that large mammals have a very strong effect on plant ecosystem structure and function. And it must also imply that large mammals are virtually immune to predators, so no trophic cascade can occur to prevent plant overgrazing.
I appreciate that it is very difficult to test such a statement since evolution has been going on for a long time before humans arrived, and so there must have been a lot of other factors causing ecosystem changes in those early years. Humans have a disproportionate love for biodiversity that is larger than us. So, we revel in elephants, tigers, bears, and whales, while at the same time we pay little attention to the insects, small mammals, most fish, and plankton. Because of this size bias, we are greatly concerned with the conservation of large animals, as we should be, but much less concerned about what is happening to the small chaps.
What is the evidence that large mammals and birds have a disproportionate influence on ecosystem structure and function? In my experience, I would say there is very little evidence for strong ecosystem effects from the collapse of the megafauna. DeMaster et al. (2006) evaluated a proposed explanation for ecosystem collapse caused by whaling in the North Pacific Ocean and concluded that the evidence was weak for a sequential megafauna collapse caused by commercial whaling. Trites et al. (2007) and Wade et al. (2007) supported this conclusion. Citing paleo-ecological data for Australia, Johnson (2010) and Rule et al. (2012) argued in another evaluation of ecosystem changes that the human-driven extinction of the megafauna in Australia resulted in large changes in plant communities, potentially confounded by climate change and increases in fire frequency about 40K years ago. If we accept these controversies, we are left with trying to decide if the current losses of large mammals are of similar strength to those assigned to the Pleistocene megafauna, as suggested by Smith et al. (2018).
If we define ecosystem function as primary productivity and ecosystem structure as species diversity, I cannot think of a single case in recent studies where this idea has been clearly tested and supported. Perhaps this simply reflects my biased career working in arctic and subarctic ecosystems in which the vast majority of the energy flow in the system rotates through the smaller species rather than the larger ones. Take the Great Plains of North America with and without the bison herds. What aspect of ecosystem function has changed because of their loss? It is impossible to say because of human intervention in the fire cycle and agricultural pre-emption of much of the landscape. It is certainly correct that overgrazing impacts can be severe in human-managed landscapes with overstocking of cattle and sheep, and that is a tragedy brought on by economics, predator elimination programs, and human land use decisions. All the changes we can describe with paleo-ecological methods have potential explanations that are highly confounded.
I think the challenge is this: to demonstrate that the loss of large mammals at the present time creates a large change in ecosystem structure and function with data on energy flow and species diversity. The only place I can see it possible to do this experimentally today would be in arctic Canada where, at least in some areas, caribou come and go in large numbers and with relatively little human impact. I doubt that you could detect any large effect in this hypothetical experiment. It is the little chaps that matter to ecosystem function, not the big chaps that we all love so much. And I would worry if you could do this experiment, the argument would be that it is a special case of extreme environments not relevant to Africa or Australia.
No one should want the large mammals and birds to disappear, but the question of how this might play out in the coming 200 years in relation to ecosystem function requires more analysis. And unlike the current political inactivity over the looming crisis in climate change, we conservation biologists should certainly try to prevent the loss of megafauna.
Choquenot, D., and Bowman, D.M.J.S. 1998. Marsupial megafauna, Aborigines and the overkill hypothesis: application of predator-prey models to the question of Pleistocene extinction in Australia. Global Ecology and Biogeography Letters 7: 167-180.
DeMaster, D.P., Trites, A.W., Clapham, P., Mizroch, S., Wade, P., Small, R.J., and Hoef, J.V. 2006. The sequential megafaunal collapse hypothesis: testing with existing data. Progress in Oceanography 68(2-4): 329-342. doi:10.1016/j.pocean.2006.02.007
Grayson, D.K. 1977. Pleistocene avifaunas and the Overkill Hypothesis. Science 195: 691-693.
Guthrie, R.D. 1984. Mosaics, allelochemics and nutrients: An ecological theory of late Pleistocene megafaunal extinctions. In: Quaternary Extinctions: A Prehistoric Revolution ed by P.S. Martin and R.G. Klein. University of Arizona Press Tucson.
Johnson, C.N. 2010. Ecological consequences of Late Quaternary extinctions of megafauna. Proceeding of the Royal Society of London, Series B 276(1667): 2509-2519. doi: 10.1098/rspb.2008.1921.
Martin, P.S., and Wright, H.E. (eds). 1967. Pleistocene Extinctions; The Search for a Cause. Yale University Press, New Haven, Connecticut. 453 pp.
Rule, S., Brook, B.W., Haberle, S.G., Turney, C.S.M., Kershaw, A.P., and Johnson, C.N. 2012. The aftermath of megafaunal extinction: ecosystem transformation in Pleistocene Australia. Science 335(6075): 1483-1486. doi: 10.1126/science.1214261.
Smith, F.A., Elliott Smith, R.E., Lyons, S.K., and Payne, J.L. 2018. Body size downgrading of mammals over the late Quaternary. Science 360(6386): 310-313. doi: 10.1126/science.aao5987.
Trites, A.W., Deecke, V.B., Gregr, E.J., Ford, J.K.B., and Olesiuk, P.F. 2007. Killer whales, whaling, and sequential megafaunal collapse in the North Pacific: a comparative analysis of the dynamics of marine mammals in Alaska and British Columbia following commercial whaling. Marine Mammal Science 23(4): 751-765. doi: 10.1111/j.1748-7692.2006.00076.x.
Wade, P.R., et al. 2007. Killer whales and marine mammal trends in the North Pacific – a re-examination of evidence for sequential megafaunal collapse and the prey-switching hypothesis. Marine Mammal Science 23(4): 766-802. doi: 10.1111/j.1748-7692.2006.00093.x.